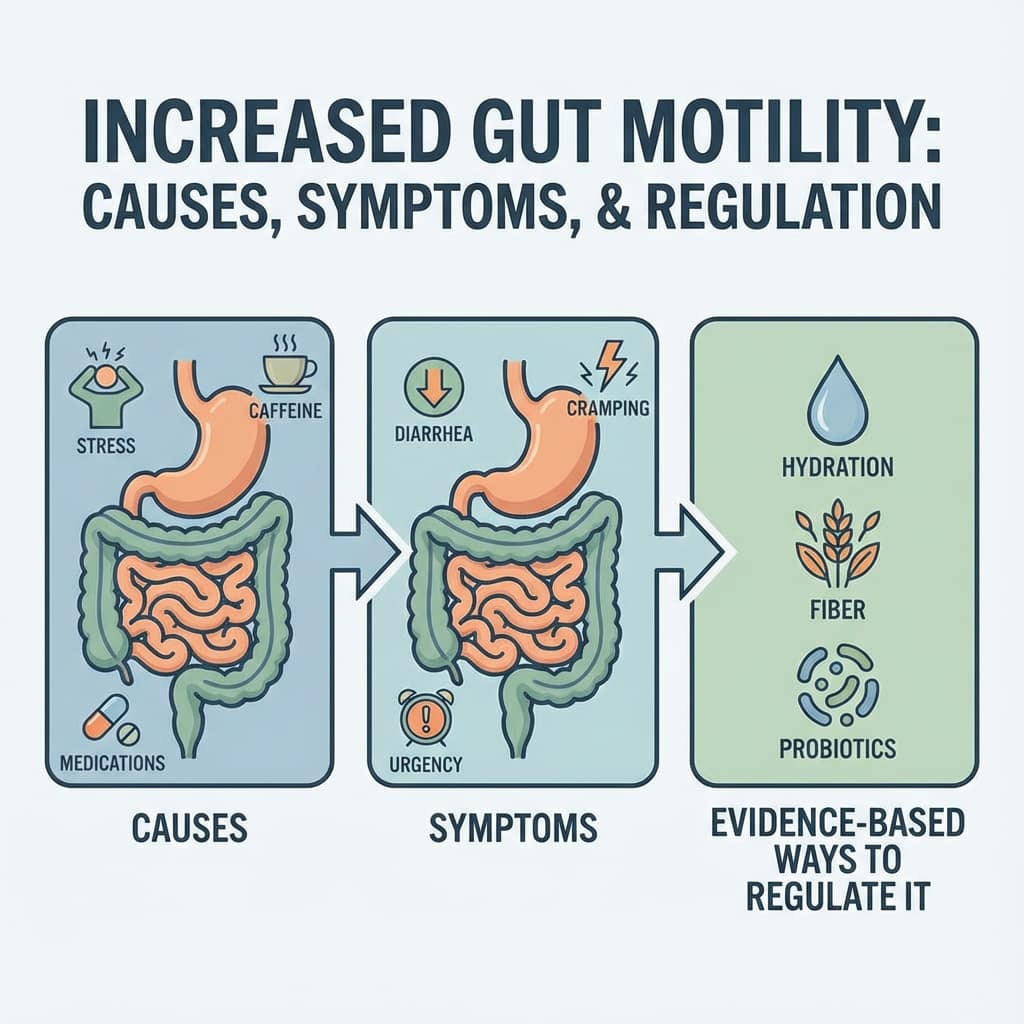
Quick answer: what “increased gut motility” means and why it matters
Increased gut motility is exactly what it sounds like—your digestive system hitting the accelerator when it should be cruising at a reasonable speed. Motility refers to the rhythmic contractions of the digestive muscles that move food through the GI tract, playing a crucial role in the normal functioning of your gastrointestinal system. In simple terms, it means the contents of your gastrointestinal tract are moving faster than normal, often leading to diarrhea, cramping, and that dreaded urgency that has you mapping out every bathroom between home and wherever you’re going.
Here’s the context that matters: normal whole gut transit time—the journey from mouth to exit—typically takes around 30 to 40 hours in healthy adults. When motility kicks into overdrive, that transit time can drop to under 20 to 24 hours, and sometimes even faster. Clinical studies measuring gut transit time consistently show this range as the threshold where things start going sideways (and frequently).
What’s important to understand is that increased gut motility isn’t a disease itself. It’s a physiological change—a symptom that shows up across a wide range of conditions:
- Acute infections (the classic food poisoning scenario)
- Irritable bowel syndrome with diarrhea (IBS-D)
- Inflammatory bowel disease flares
- Hyperthyroidism revving up everything, including your gut
- Anxiety and stress turning your digestive system into a stress ball
Now, flip the coin. Decreased motility looks like the opposite problem: infrequent bowel movements, straining, and constipation symptoms that make bathroom visits an ordeal rather than a relief. Patients with constipation often deal with slow transit constipation or functional constipation, where the digestive tract moves contents at a glacial pace.
The key takeaway: your gastrointestinal system works best in a Goldilocks zone—not too fast, not too slow. Both extremes can cause real problems, from nutrient malabsorption to impaired gut motility affecting your entire quality of life.
How gut motility normally works
Think of your digestive tract as an incredibly sophisticated conveyor belt, except instead of mechanical rollers, it uses coordinated muscle contractions in the walls of your esophagus, stomach, small intestine, and colon to move everything along. This process—gastrointestinal motility—is what separates a functional digestive system from one that leaves you either racing to the bathroom or wondering when anything will happen at all.
The two main players in this muscular symphony are peristalsis and segmentation. Peristalsis involves wave-like contractions that push food forward—imagine squeezing a tube of toothpaste from the back. These waves propagate along the intestinal wall, propelling contents toward the exit. Segmentation, on the other hand, involves ring-like contractions that mix and churn the chyme (that lovely partially digested food paste), maximizing contact with the intestinal lining for nutrient absorption.
Normal transit times differ dramatically depending on which part of the gi tract you’re looking at:
- Stomach emptying: 2–4 hours for most solid meals
- Small intestine transit: 3–5 hours
- Colonic transit time: 20–30 hours (this is where things slow down for water absorption)
- Total whole gut transit time: 30–40 hours in healthy subjects
There’s also a housekeeping crew that works the night shift. The migrating motor complex (MMC) is a fasting pattern that sweeps through the small intestine every 90 to 120 minutes when you haven’t eaten. Think of it as the intestinal cleaning service, pushing residual debris, bacteria, and waste forward to prevent bacterial overgrowth from setting up shop where it doesn’t belong.
Healthy gi motility ensures three critical functions: nutrient absorption, microbial balance, and waste elimination. Both the enteric nervous system (that “second brain” in your gut) and input from your actual brain coordinate the timing and strength of these contractions. When this coordination works well, you barely notice it. When it doesn’t—that’s when things get uncomfortable.
Neural, hormonal, and microbial control of gut motility
Your gut doesn’t just move on its own like some mindless tube. The reality is far more complex: gi motility is orchestrated by an intricate network of interacting systems—the enteric nervous system, the autonomic nervous system, hormones, immune cells, and even your gut microbiota. All of these players work together (or sometimes against each other) in regulating gut motility.
The enteric nervous system deserves its reputation as a “second brain.” It contains somewhere between 200 and 600 million neurons organized into the myenteric and submucosal plexuses embedded in the gut wall. Classic experiments from the mid-20th century demonstrated something remarkable: isolated sections of intestine can generate peristaltic movements even when completely disconnected from the central nervous system. Your gut can literally think for itself when it comes to moving things along.
But the ENS doesn’t work in isolation. The autonomic nervous system provides top-down input. The parasympathetic branch (mainly the vagus nerve and pelvic nerves) generally stimulates motility—rest and digest mode. The sympathetic nervous system typically slows things down, redirecting blood flow away from digestion during fight-or-flight scenarios. Here’s the twist: acute stress can either speed up or slow down motility depending on the individual, which partly explains why some people get stress-induced diarrhea while others get stress-induced constipation.
Hormones and neurotransmitters add another layer of control. Serotonin (5-HT) is the superstar here—about 95% of your body’s serotonin lives in the gut, released by enterochromaffin cells when the intestinal wall stretches. This triggers peristaltic reflexes through the gut brain axis. Other key players include:
- Motilin: triggers the MMC during fasting
- Cholecystokinin: stimulates gallbladder contraction and slows gastric emptying
- Gastrin: promotes stomach acid production and gastric motility
- Peptide YY: slows intestinal transit after meals
The immune system also gets involved. When immune cells like mast cells become activated, they release mediators that can sensitize ENS neurons and alter muscle function. This explains why infections and inflammation can dramatically affect gut motility—and why that altered contractility sometimes persists long after the initial insult.
Then there’s the microbiome factor. Your gut bacteria don’t just passively sit there—they actively influence gut motility through several mechanisms. Bacterial fermentation produces short-chain fatty acids (SCFAs) like acetate, propionate, and butyrate that affect epithelial cells and neural pathways regulating motility. Human studies from the 2010s onwards have increasingly linked dysbiosis—imbalances in gut microbiota composition—to gastrointestinal motility disorders including IBS and chronic motility dysfunction.
Signs and symptoms of increased gut motility
Increased motility can technically affect any segment of your gastrointestinal tract, but you really feel it when the small intestine or colon kicks into high gear. That’s when decreased gut transit time starts causing noticeable problems.
The most common patient-reported complaints include:
- Frequent loose or watery stools
- Urgency (the “I need a bathroom NOW” feeling)
- Abdominal cramping, especially before bowel movements
- Loud bowel sounds (borborygmi)
- Bloating with rapid gas passage
- A frustrating sense of incomplete evacuation despite diarrhea
Frequent loose stools are the hallmark symptom. Increased gut motility leads to reduced gut transit time, which contributes to watery stools and malabsorption. When colonic transit speeds up, there’s simply not enough time for water reabsorption—the colon normally removes about 90% of the water from intestinal contents. Shortened residence time means watery output.
Urgency is particularly distressing. Normally, high-amplitude propagating contractions (the powerful waves that move stool toward the rectum) occur about 6 to 8 times daily. When these HAPCs become more frequent or clustered, you get that sudden, barely-controllable need to go. In severe cases, this can progress to fecal incontinence—loss of bowel control that affects millions of people but rarely gets discussed openly.
Cramping and abdominal discomfort often precede bowel movements when motility is increased. These sensations originate from the pelvic floor muscles and intestinal smooth muscle contracting more forcefully than usual. The pelvic floor can also become dyscoordinated, adding to the discomfort.
Malabsorption is a serious consequence when increased motility becomes chronic. Rapid transit means nutrients don’t have adequate contact time with the intestinal lining. This can lead to:
- Weight loss
- Fatigue
- Iron deficiency
- B12 deficiency
- Fat-soluble vitamin deficiencies (A, D, E, K)
Red flags that demand immediate attention:
When increased motility comes with certain warning signs, it’s no longer a wait-and-see situation. Nocturnal diarrhea that wakes you from sleep, blood or mucus in stool, fever, severe pain, anemia, or unintentional weight loss exceeding 5% of body weight over 6 to 12 months all suggest something more serious than a functional problem.
Finally, many people oscillate between increased and decreased motility—a pattern seen in IBS mixed type. One week you’re dealing with urgency and loose stools; the next, you’re straining and dealing with constipation symptoms. This variability makes self-diagnosis unreliable, which is why tracking symptom patterns over several weeks provides much more useful information for healthcare providers.
Why gut motility increases: common causes and mechanisms
Increased gut motility can be a brief blip (think: anxiety before a big presentation) or a chronic problem (think: years of dealing with IBS-D). Often, multiple overlapping triggers converge to push the digestive system into overdrive. Understanding these risk factors helps target the right solutions.
Infectious causes
Acute viral or bacterial gastroenteritis remains the most common cause of sudden-onset increased motility. Pathogens like norovirus, Salmonella, and Clostridioides difficile release toxins that trigger massive secretion and speed colonic transit. The result: high-volume, watery diarrhea that’s actually your body’s attempt to flush out the invaders. Most resolve within days, but some leave lasting changes—more on that later.
Functional disorders: irritable bowel syndrome
Irritable bowel syndrome ibs—particularly the diarrhea-predominant type (IBS-D)—represents the prototype of abnormal motility without visible structural damage. Rome IV criteria, published in 2016, standardized the diagnosis: recurrent abdominal pain associated with defecation or changes in stool frequency/form, present at least one day per week for three months.
What’s actually happening? Altered brain–gut signaling, visceral hypersensitivity (normal contractions feel painful), and microbiome shifts all contribute. Interestingly, giant migrating contractions in the lower small intestine—normally silent—often correlate with abdominal pain in IBS patients. The contractions themselves may be normal in force; it’s the sensory processing that’s amplified.
Functional dyspepsia, affecting up to 20% of adults in North America, shares some of these mechanisms, though it primarily affects the upper gi tract with symptoms like early satiety, bloating, and upper abdominal pain.
Inflammatory bowel disease
Crohn’s disease and ulcerative colitis cause chronic inflammation that directly impacts gut motility. Mucosal inflammation, ulceration, and sometimes intestinal obstruction create abnormal motility patterns—often with urgency, frequent stools, blood, and mucus. IBD requires specific treatment beyond motility management.
Endocrine and metabolic causes
Hyperthyroidism is notorious for speeding up everything, including intestinal transit. Diabetes can cause autonomic neuropathy affecting the digestive tract—paradoxically sometimes causing rapid transit despite the more commonly discussed gastroparesis (delayed gastric emptying). Carcinoid syndrome, where neuroendocrine tumors overproduce serotonin, causes flushing and explosive diarrhea.
Diet and medication triggers
What goes in directly affects how fast it comes out:
- High caffeine doses: stimulate colonic motility
- Alcohol: irritates the gut lining and speeds transit
- Sugar alcohols (sorbitol, mannitol): osmotically active, draw water into the colon
- High-FODMAP foods: fermentable carbs that produce gas and may trigger diarrhea in sensitive individuals
- Magnesium-containing antacids and supplements: classic cause of osmotic diarrhea
Medications also impact gut motility significantly. Erythromycin acts as a motilin receptor agonist—it literally mimics the hormone that triggers intestinal housekeeping waves. Prokinetic drugs like prucalopride, metformin, some chemotherapy agents, and various antibiotics can all accelerate transit.
Psychological factors
Pre-exam diarrhea. Runner’s trots. That churning feeling before a difficult conversation. The gut brain axis is real, and acute stress activates autonomic and ENS responses that can dramatically speed up (or sometimes paradoxically slow down) motility. Chronic anxiety and stress perpetuate these patterns.
Some people also experience acid reflux or gastroesophageal reflux disease symptoms alongside their motility issues—the two often coexist because both involve dysfunctional neuromuscular control of the upper gi tract, sometimes accompanied by chest pain or difficulty swallowing.
Immune system, increased motility, and diarrhea
When your immune system detects a threat in the gut, it doesn’t politely ask it to leave. It mounts a response that often includes speeding up motility to flush out whatever shouldn’t be there. This makes evolutionary sense—but the same mechanisms can malfunction in chronic conditions.
Acute intestinal infections trigger rapid cytokine release. Pro-inflammatory molecules like TNF-α, IL-1β, and IL-6 signal danger, while mast cell activation amplifies the response. The combined effect: increased chloride and water secretion into the intestinal lumen plus enhanced propulsive contractions. Water pours in, motility speeds up, and diarrhea ensues. It’s an aggressive but effective defense against pathogens.
The problem arises when chronic inflammation takes hold. In inflammatory bowel disease, this immune activation persists, driving ongoing symptoms. But even in conditions without visible inflammation, subtle immune changes can perpetuate increased motility.
Post-infectious IBS is a prime example. After acute gastroenteritis resolves, some patients develop lasting IBS symptoms—typically diarrhea-predominant. Studies show that even when visible inflammation disappears, microscopic immune changes and altered nerve signaling persist. The initial infection essentially rewires the gut’s response patterns.
Research from the 2000s and 2010s documented increased mast cells near ENS nerve fibers in some IBS-D patients. These sensitized immune cells may explain why stress, certain foods, or hormonal changes can trigger symptom flares—any stimulus that activates these primed mast cells can set off cramping and urgent bowel movements.
The immune system also mediates motility changes in conditions originating outside the gut. Celiac disease causes immune-mediated damage to the small intestinal lining, affecting absorption and motility. Systemic sclerosis can damage smooth muscle and nerves throughout the digestive tract. These autoimmune conditions highlight how closely gut function depends on balanced immune regulation.
Diet, microbiota, and increased gut motility
What you eat doesn’t just become nutrients—it shapes your gut environment and directly affects how fast (or slow) everything moves through. Diet-induced changes in the gut microbiome and luminal chemistry are major drivers of motility patterns.
Foods that speed things up
Certain dietary patterns consistently associate with faster transit:
- Very high FODMAP intake: Fermentable Oligo-, Di-, Mono-saccharides And Polyols ferment rapidly, producing gas and drawing water into the colon
- Spicy foods: Capsaicin stimulates motility receptors
- Large caffeine doses: Coffee is a known colonic stimulant
- High-fat fast-food meals: Trigger strong gastrocolic reflexes
- Excessive alcohol: Irritates mucosa and accelerates transit
In contrast, moderate fermentable fiber, balanced meals, and adequate (but not excessive) fluid tend to produce more formed, regular bowel movements in most people.
Sugar alcohols: the hidden culprits
Sorbitol, mannitol, xylitol, and other sugar alcohols lurk in “sugar-free” products, gums, candies, and some medications. They’re also naturally present in fruits like prunes, apples, and pears. These compounds are poorly absorbed in the small intestine, so they reach the colon intact where they draw water osmotically and undergo fermentation. In high amounts, they reliably cause diarrhea—manufacturers are required to add warning labels about “laxative effects” on products exceeding certain thresholds.
Fermentable fibers: a double-edged sword
Fermentable fibers like inulin and fructans increase SCFA production and gas. For some people, this gently promotes healthy motility. For IBS-D patients with visceral hypersensitivity, it can precipitate urgency and diarrhea. The difference lies in individual tolerance and gut microbiome composition.
Dysbiosis and motility
Your gut microbiome isn’t just along for the ride—it actively modulates motility through SCFA production, bile acid metabolism, and interactions with immune cells. Dysbiosis—loss of bacterial diversity, reduced Bifidobacterium and Lactobacillus, overgrowth of pro-inflammatory species—correlates with altered motility patterns. Changes in gut ph from fermentation products can further influence the environment.
Small intestinal bacterial overgrowth (SIBO) represents an extreme example. When bacteria proliferate abnormally in the upper small intestine, they consume nutrients before absorption, produce gas, and trigger bloating, pain, and diarrhea soon after eating. Paradoxically, while SIBO often develops from slow motility (allowing bacterial buildup), the resulting fermentation can provoke defensive increases in motility.
Bile acid diarrhea: the under-recognized problem
Bile acid malabsorption deserves special mention as an often-overlooked cause of chronic diarrhea. Normally, bile acids released to digest fat are reabsorbed in the ileum. When this reabsorption fails—after ileal resection, in some IBS-D cases, or with certain genetic variations—excess bile acids reach the colon. There, they stimulate secretion and motility directly. Studies suggest bile acid diarrhea may account for up to 25-30% of IBS-D cases, yet it’s frequently missed without specific testing.
Immediate impact of physical activity on gut motility
Here’s something that might affect your pre-workout bathroom strategy: physical activity can change motility patterns almost immediately—sometimes beneficially, sometimes inconveniently.
A 2025 study published in Scientific Reports (Katagiri et al.) examined the immediate effect of physical activity on gut motility in healthy adults. The setup was straightforward: 21 participants walked on a treadmill for approximately 3,000 steps while researchers monitored bowel sounds—a non-invasive proxy for peristaltic activity.
The findings were striking. Bowel sound indices increased within just 1 to 2 minutes after starting to walk, then returned to baseline by about 3 minutes post-exercise. This suggests that even moderate walking transiently boosts motility.
Several mechanisms likely explain this effect:
- Autonomic nervous system modulation: Exercise shifts autonomic balance, initially stimulating sympathetic activity but ultimately promoting parasympathetic tone during recovery
- Splanchnic blood flow redistribution: Movement affects blood flow to the gut
- Mechanical oscillation: Physical jostling of abdominal contents may stimulate local reflexes in the intestinal wall
For people dealing with sluggish transit, this motility boost is good news. Regular physical activity consistently helps normalize bowel movements in patients with constipation and anorectal dysfunction.
But there’s a flip side. Intense or prolonged exercise—particularly distance running—can tip some people into cramping and urgent loose stools. “Runner’s diarrhea” is well-documented in endurance athletes, likely reflecting prolonged gut blood flow reduction, mechanical stress, and altered autonomic regulation during extended exertion.
The practical takeaway: moderate activity generally supports healthy human colonic motility, but individual responses vary. If you’re prone to exercise-induced symptoms, experimenting with timing, intensity, and pre-workout nutrition can help find your sweet spot.
When increased gut motility is dangerous: red flags and urgent evaluation
Most episodes of increased motility are benign. A bout of traveler’s diarrhea, stress-induced urgency, or caffeine-triggered loose stools typically resolve without drama. But certain patterns demand prompt medical evaluation.
New-onset diarrhea with visible blood, tarry stools, or pus
Blood in stool is never normal. Whether bright red (suggesting lower GI bleeding) or dark and tarry (suggesting upper GI bleeding), this requires evaluation. Pus or mucus points toward inflammatory or infectious processes.
Diarrhea with fever or severe abdominal pain
The combination suggests infection, acute inflammation, or intestinal obstruction. This isn’t a “wait it out” situation.
A rare but serious motility disorder to be aware of is intestinal pseudo obstruction. This condition can mimic other gastrointestinal issues like gastroparesis, but it may be life-threatening. Intestinal pseudo obstruction involves impaired gut motility due to nerve or muscle dysfunction within the enteric nervous system, and requires urgent medical attention.
Unintentional weight loss
Losing more than 5% of body weight over 6 to 12 months without trying signals something is wrong—malabsorption, malignancy, or other serious gastrointestinal disorders.
Nocturnal diarrhea
Diarrhea that wakes you from sleep suggests organic disease rather than functional causes. Your gut should be relatively quiet overnight.
Progressive anemia
Chronic blood loss or malabsorption can cause anemia, manifesting as fatigue, pallor, and sometimes shortness of breath.
Recent antibiotic use with watery diarrhea
This pattern raises immediate concern for Clostridioides difficile colitis—a potentially serious infection requiring specific treatment.
Recent travel with prolonged symptoms
Symptoms lasting more than two weeks after travel to high-risk regions warrant evaluation for parasites and other endemic infections.
Time thresholds matter
Clinical practice distinguishes:
- Acute diarrhea: Less than 14 days—often self-limited
- Persistent diarrhea: 14–30 days—warrants investigation
- Chronic diarrhea: More than 4 weeks—always requires evaluation
People with personal or family history of inflammatory bowel disease, colorectal cancer, or celiac disease should have a lower threshold for seeking evaluation when experiencing persistent increased motility.
Typical workup
Achieving an accurate diagnosis for chronic or concerning symptoms generally involves:
Test Type | Purpose |
|---|---|
Blood tests (CBC, CRP, thyroid function) | Screen for anemia, inflammation, hyperthyroidism |
Stool tests (pathogens, calprotectin, occult blood) | Identify infection and inflammation |
Celiac serology | Rule out celiac disease |
Imaging or colonoscopy | Assess for structural disease, especially with red flags or age ≥45-50 |
A definitive diagnosis often requires combining clinical history with targeted testing, particularly when mild symptoms persist without obvious cause.
Evidence-based strategies to normalize increased gut motility
Before diving into solutions, an important caveat: treatment depends entirely on the underlying cause. Self-treatment shouldn’t delay proper evaluation when red flags are present. That said, once serious conditions are ruled out, several evidence-based strategies can help restore more normal motility.
Dietary strategies
Low-FODMAP diet: Developed in the late 2000s and now supported by guidelines for IBS management, this structured elimination and reintroduction approach identifies personal trigger foods. It’s meant to be done with dietitian supervision—not as a permanent restriction but as a diagnostic tool.
Caffeine and alcohol moderation: Both stimulate motility. Cutting back often provides noticeable benefit.
Sugar alcohol awareness: Check labels on “sugar-free” products. Reducing sorbitol and similar compounds can quickly reduce osmotic diarrhea.
Moderate soluble fiber: During active diarrhea, very high dietary fiber intake can worsen symptoms. Moderate soluble fiber (oats, psyllium) may help normalize stool consistency without adding bulk that speeds transit.
Mindful fat intake: Large high-fat meals trigger strong gastrocolic reflexes. Smaller, more frequent meals may reduce post-meal urgency.
Nutritional supplements may be needed if malabsorption has caused deficiencies—iron, B12, vitamin D, and other fat-soluble vitamins commonly require repletion.
Pharmacologic options
Medication | Indication | Notes |
|---|---|---|
Loperamide | Acute non-bloody diarrhea | Slows motility, OTC available |
Bile acid binders (cholestyramine) | Bile acid diarrhea | Often dramatically effective when diagnosis confirmed |
Antispasmodics (hyoscine butylbromide) | Cramping | Target muscle contractions directly |
5-HT3 antagonists (alosetron) | Severe IBS-D (selected cases) | Restricted prescribing due to safety concerns |

Microbiota-directed approaches
Strain-specific probiotics show emerging evidence for certain types of diarrhea. Lactobacillus and Saccharomyces boulardii strains have demonstrated benefit in antibiotic-associated diarrhea prevention through randomized controlled trial data. However, evidence for IBS-D and chronic functional diarrhea remains mixed and highly strain-specific. Not all probiotics are interchangeable.
For documented small intestinal bacterial overgrowth, targeted antibiotics (notably rifaximin) can address the underlying bacterial overgrowth driving symptoms. Further research continues to clarify which patients benefit most.
Lifestyle and stress management
Regular moderate physical activity: As discussed, movement generally supports healthy motility—with monitoring for those prone to exercise-induced symptoms.
Psychological therapies: Cognitive-behavioral therapy and gut-directed hypnotherapy (pioneered in the 1980s but refined since) can reduce brain–gut axis hyperreactivity. Biofeedback therapy helps some patients, particularly those with pelvic floor dysfunction contributing to symptoms.
Relaxation techniques: Given the gut brain axis connection, reducing chronic stress through meditation, breathing exercises, or other methods can meaningfully impact motility patterns.
Combination approaches
In practice, effective treatment plans often combine strategies. For example, an IBS-D patient might follow a low-FODMAP diet, use loperamide as needed for breakthrough symptoms, engage in gut-directed hypnotherapy, and take a targeted medication based on their specific symptom profile. This systematic review-informed approach tends to outperform any single intervention.
The key is collaboration with healthcare professionals who can help tailor the approach to your specific diagnosis and response patterns.
Research frontiers: from motility testing to targeted therapies
Modern motility research has moved far beyond simply categorizing problems as “too fast” or “too slow.” Advanced tools now dissect specific patterns of human colonic motility and small bowel function, enabling increasingly targeted treatments.
Assessment methods
Whole-gut transit studies use radio-opaque markers or wireless motility capsules to objectively measure transit time through each segment. High-resolution manometry provides detailed pressure mapping in the esophagus and anorectum. Breath tests detect small intestinal bacterial overgrowth and carbohydrate malabsorption. Magnetic resonance imaging offers radiation-free visualization of gut function in motion.
These tools help distinguish between different types of gastrointestinal motility disorders and guide treatment selection.
Emerging therapeutic targets
Research continues to explore:
- Serotonergic signaling pathways: New 5-HT modulators targeting motility without central nervous system effects
- Bile acid receptors (FXR, TGR5): Promising targets for bile acid-related diarrhea
- Nerve-immune-microbiota interactions: Understanding how these systems crosstalk may unlock new interventions
Post-2010 clinical trials have examined rifaximin for IBS-D, novel bile acid modulators, and even microbiota transplants for selected conditions. Precision medicine approaches aim to match therapies to individual motility patterns, gut microbiome profiles, and immune system signatures.
What we still need to learn
Despite progress, significant gaps remain. A systematic review of small intestinal motility in common diarrheal conditions would likely reveal how little detailed human data exists compared to colonic motility research. Longitudinal studies linking specific microbiota configurations, SCFA profiles, and immune markers to measured transit times and symptom trajectories in IBS-D, IBD, and post-infectious diarrhea are still needed.
The field is moving toward understanding gastrointestinal gi motility as a personalized phenomenon—where your specific pattern of gut function, microbial composition, and neural sensitivity determines the best intervention rather than one-size-fits-all treatment.
Summary
Increased gut motility is common, complex, and arises from the interplay of neural, hormonal, immune, microbial, and lifestyle factors. Whether it’s a brief episode of pre-interview jitters or a chronic pattern affecting your daily life, understanding what’s driving your digestive system helps distinguish between benign, self-limited episodes and conditions requiring thorough evaluation.
Diet, medications, physical activity, and stress management all influence gut motility—but the most effective approaches are tailored to specific diagnoses and individual responses. What works for one person may not work for another, and that’s okay. The goal is finding your personal formula for healthy subjects and healthy digestion.
If you’re experiencing persistent or severe symptoms—especially red flags like blood, weight loss, fever, or nocturnal diarrhea—document your patterns and consult a healthcare professional. Your gut is trying to tell you something. It’s worth listening.
Diagnosis of increased gut motility
Diagnosing increased gut motility is a crucial step in managing symptoms and improving quality of life, especially since gastrointestinal motility disorders often present with overlapping complaints like diarrhea, urgency, or abdominal discomfort. Because gut motility can be influenced by a wide range of factors—from irritable bowel syndrome (IBS) to chronic constipation, inflammatory bowel disease, and even small intestinal bacterial overgrowth (SIBO)—an accurate diagnosis is essential for effective treatment.
The process begins with a thorough medical history. Your healthcare provider will ask about the frequency and consistency of your bowel movements, the presence of abdominal pain or bloating, and any changes in your digestive tract function. They’ll also want to know about risk factors such as recent travel, medication use, dietary habits, and any family history of gastrointestinal disorders. A physical examination may include gentle palpation of the abdomen to check for tenderness, masses, or signs of intestinal obstruction, as well as an assessment of the pelvic floor muscles and sphincter muscles for strength and coordination—especially if symptoms like fecal incontinence or difficulty with evacuation are present.
To pinpoint the underlying cause of abnormal motility, several diagnostic tests may be used:
- Gastrointestinal transit study: This test tracks the movement of food or markers through your digestive tract, helping to identify patterns of slow transit constipation or, conversely, accelerated gut motility.
- Anorectal manometry: By measuring the pressure and function of the anal sphincter and pelvic floor muscles, this test can reveal issues contributing to fecal incontinence or functional constipation.
- Colonic transit study: Focused on the large intestine, this test helps determine whether slow or rapid movement through the colon is responsible for your symptoms.
- Gastric emptying study: This evaluates how quickly food leaves the stomach, which is important for diagnosing conditions like gastroparesis or, less commonly, abnormally rapid gastric emptying.
- Magnetic resonance imaging (MRI): Advanced imaging can visualize the structure and function of the gastrointestinal tract, helping to rule out inflammatory bowel disease, intestinal obstruction, or other structural abnormalities.
- Wireless motility capsule: This innovative test measures pressure, pH, and temperature as it travels through your GI tract, providing a comprehensive picture of motility from stomach to colon.
A definitive diagnosis of increased gut motility relies on integrating your symptoms, medical history, and the results of these specialized tests. This comprehensive approach helps distinguish between different gastrointestinal motility disorders and guides the selection of targeted therapies—whether that means dietary changes, nutritional supplements, medications, or, in rare cases, surgical intervention.
It’s important to remember that increased gut motility is often a symptom rather than a standalone disease. Conditions like irritable bowel syndrome (IBS), chronic constipation, and small intestinal bacterial overgrowth can all impact gut motility in different ways. That’s why an accurate diagnosis is so important: it ensures you receive the right treatment for your specific situation.
In addition to medical interventions, lifestyle modifications play a key role in managing motility disorders. Eating a balanced diet rich in dietary fiber, staying well-hydrated, engaging in regular physical activity, and managing stress through techniques like meditation or yoga can all support healthy GI motility and improve overall gut function. Avoiding trigger foods that exacerbate symptoms is also a practical step for many patients.
As research continues to evolve, systematic reviews and further studies will help refine our understanding of the best diagnostic tools and treatment strategies for increased gut motility. For now, working closely with your healthcare provider to achieve an accurate diagnosis remains the cornerstone of effective care.